
Pharmaceuticals
How is Innovation and Technology Reshaping the India CDMO Sector?
October 11, 2024
by Ken ResearchThe Indian Contract Development and Manufacturing Organization (CDMO) market is set to redefine the global landscape. With a current valuation of USD 3,256.9 million and projected to double by 2028, the sector is projected to outpace global growth rates, boasting a rate (CAGR) of 11.6%. This is not a mere statistical insight but a strategic shift driven by strong regulations, progressive government reforms, continuous technological advancements, and extensive investments.
In this article, we take you through the key drivers behind this transformation, highlighting the role of regulatory frameworks, technological innovations, and strategic investments. India is on the brink of strengthening its position as the foremost hub for contract development and manufacturing, setting new benchmarks for the industry.
At a Glance
1. Government Regulations and Reforms: Firm obedience to Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP) by CDSCO ensures high standards.
2. Automation and Digital Transformation: One Lab’, a digital platform contributes to the reduction in compliance risk through the standardization of process and improved data quality.
3. Expansion of Manufacturing Capabilities: Talent retention strategies have reduced turnover rates by 20%, maintaining project continuity and quality.
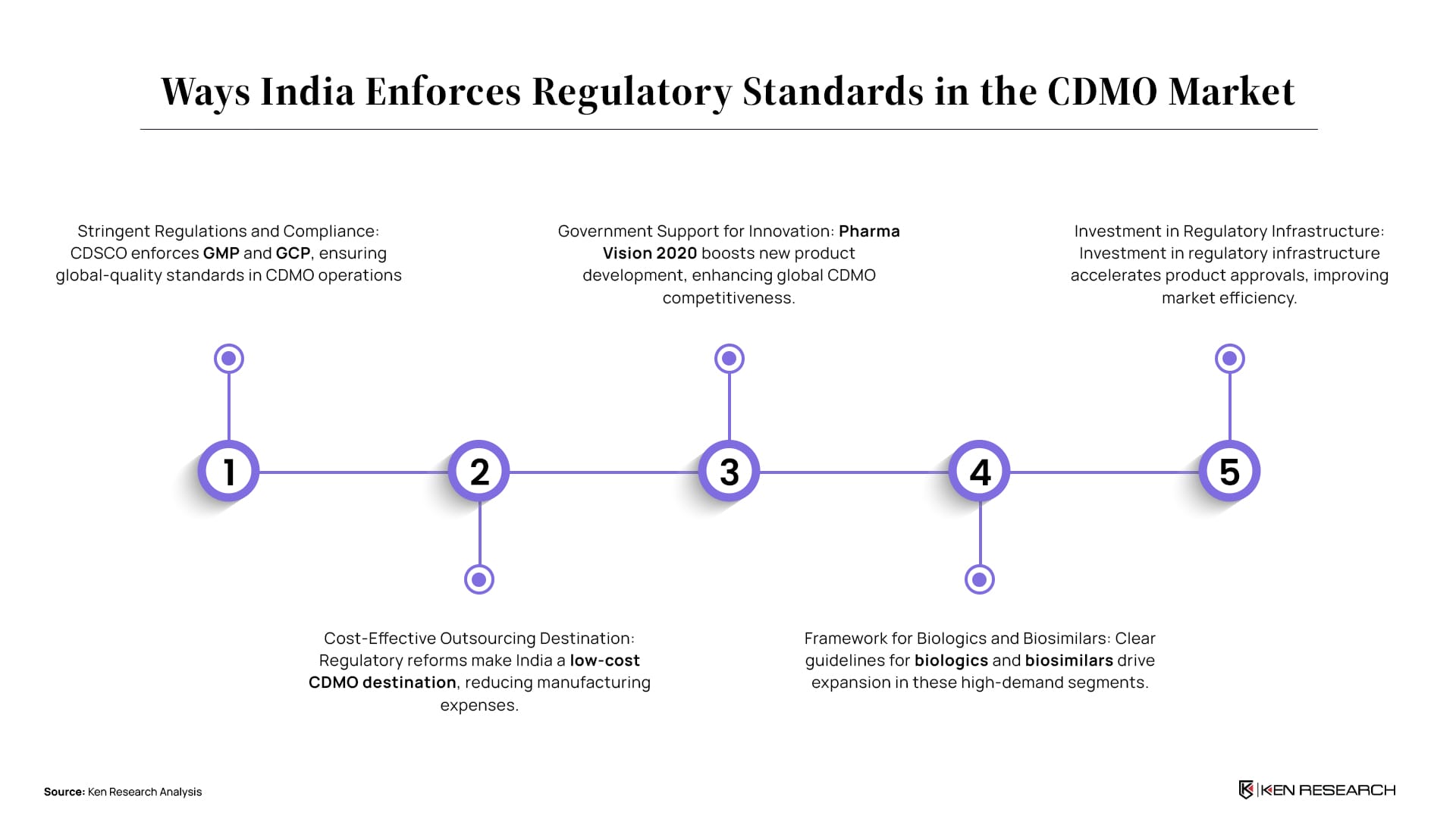
India Enforces Standards in CDMO Market
The Regulatory Board in India's CDMO market plays an important role in enforcing quality standards, ensuring patient safety and drug efficacy. Government reforms can incentivize CDMO growth by simplifying approval processes, offering tax breaks, and supporting R&D investment. This creates a more attractive environment for domestic and international CDMO players.
1. Stringent Regulations Ensure Quality and Compliance: The Central Drugs Standard Control Organization (CDSCO) mandates adherence to Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP), ensuring high-quality standards.
Catalent has utilized India's strong regulatory framework to ensure compliance, attracting global clients and contributing to market growth.
2. Favorable Regulatory Environment for Outsourcing: India’s regulatory reforms have made it a cost-effective outsourcing destination, reducing manufacturing costs.
These cost reductions have solidified India’s position as a preferred outsourcing hub for pharmaceutical companies.
3. Regulatory Support for New Product Development: The "Pharma Vision 2020" plan by the Indian government supports new product development, boosting global competitiveness.
This initiative aims to make India a leader in the end-to-end drug manufacturing and innovation.
4. Regulatory Framework for Biologics and Biosimilars: India's clear regulatory guidelines for biologics and biosimilars drive growth in these emerging areas.
Lonza's investment in Capsugel expanded its biologics capabilities, leveraging India's regulatory clarity.
5. Investment in Regulatory Infrastructure: Several investments have been made to streamline the approval process for new products, enhancing market efficiency.
Improved regulatory infrastructure has cut approval times, hence increasing responsiveness.
India’s CDMOs Enhance Quality with Tech
India's CDMO tech ops teams are key to streamlining production and staying competitive. They implement automation, data analytics, and new technologies to boost efficiency and innovation. This allows Indian CDMOs to offer high-quality, cost-effective drug development and manufacturing.
1. Automation and Digital Transformation: Adoption of automation and digital technologies smoothen processes and boost operational efficiency.
One Lab’, a digital platform contributes to the reduction in compliance risk through the standardization of process and improved data quality.
2. Adoption of Emerging Technologies: Investments in gene and cell therapies and biologics are fulfilling growing biopharma demands.
This has led to an increase in contract acquisitions from biopharma companies.
3. Collaboration and Knowledge Sharing: CDMOs are partnering with tech providers and research institutions to drive innovation.
Collaborative efforts have increased industry knowledge sharing and fostering innovation.
4. Talent Development: Upskilling initiatives ensure expertise in complex projects and leverage the latest technologies.
Studies show that companies that invest in training programs experience higher revenue per employee, increased profit margins, and improved market share
5. Operational Efficiency: Advanced technologies and digital solutions are reducing costs and enhancing competitiveness.
Digital transformation initiatives have reduced operational costs increasing global competitiveness.
The Road to Global CDMO Leadership: India’s Strategic Plan
To become a global leader in the CDMO market, India needs to focus on continuous regulatory support, expanding capabilities, diversifying services, developing talent, and leveraging technology.
1. Continued Regulatory Support: Ongoing investment in infrastructure and efficient approval processes will support market growth.
Such investments are expected to boost market growth by an additional percentage annually.
2. Expansion of Manufacturing Capabilities: Expanding manufacturing and R&D facilities is required to meet global demand.
A global lighting company, is evaluating expanding its manufacturing capacity in India as part of a "China Plus-One" strategy.
3. Diversification of Service Offerings: Focusing on biologics, gene and cell therapies, and personalized medicines will keep Indian CDMOs competitive.
Gene therapy is a rapidly advancing field in India, with the government issuing national guidelines for gene therapy product issuing national guidelines for gene therapy product development and clinical trials in 2019.
4. Talent Development and Retention: Developing and retaining a skilled workforce ensures high-quality outcomes and competitive advantage.
Talent retention strategies have reduced turnover rates by 20%, maintaining project continuity and quality.
5. Investing in Cutting-Edge Technology: Investing in digital transformation and cutting-edge technologies will add to collaboration and innovation.
According to a survey, the technology investments are most likely to improve business operations in the next two years are automation technologies with 45%, SaaS with 35% and big data analytics at 40%.

Case Studies: How Countries Became Market Leaders
Case Study– USA
Catalent, USA were on a mission to strengthen the capabilities of their gene therapy to meet the growing demands for advanced therapeutic solutions. The primary challenge was to make sure that while expanding their services they still stick to the global regulatory requirements.
Result: Catalent acquired Paragon Bio Services, a leading in gene therapy CDMO which strengthened India’s framework while adhering to Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP) standards.
Case Study- Switzerland
Lonza a city in Switzerland was dealing with the issue of an expanding dosage form capability to serve the growing demand and deliver innovative drug delivery solutions. Their challenge was to navigate the regulatory requirements and understand the market dynamics associated with biologics and advanced drug formulations.
Result: Lonza acquired Capsugel, a leading provider of capsule-based drug delivery solutions. This acquisition was facilitated by India's well-defined regulatory framework, which provided clear guidelines for biologics and biosimilars.
Case Study– Switzerland
Siegfried needed to improve its regional capabilities to strengthen its position in the global CDMO market and meet the rising demand for pharmaceutical manufacturing services. The challenge was to effectively utilize local talent and infrastructure while ensuring compliance with international quality and regulatory standards. This required significant investment in local capabilities and aligning them with global operational standards.
Result: Siegfried invested in Alchemy Pharmatech, while utilizing India’s talented workforce and regulatory infrastructure. This strategic investment enabled Siegfried to enhance its regional capabilities, streamline operations, and achieve greater market penetration.
The Growth Trajectory of India's CDMO Industry: What Lies Ahead
Continued Growth Trajectory: The market is expected to maintain a CAGR of 8% from 2024 to 2029.
This growth will surpass the global CDMO market significantly.
Expansion into Emerging Therapies: Investments in biologics, gene, and cell therapies will capitalize on growing demand.
This strategic focus will position Indian CDMOs at the forefront of innovation.
Increased Outsourcing by Global Pharma Companies: India's cost advantages and skilled talent will attract more outsourcing from global pharmaceutical companies.
Increased outsourcing is expected to drive significant market expansion.
Technological Advancements and Digital Transformation: Ongoing investments in digital technologies will enhance operational efficiency and innovation.
Technological advancements will also improve sustainability and energy efficiency.
Consolidation and Strategic Partnerships: Increased consolidation and partnerships will strengthen market capabilities and global reach.
Strategic collaborations have improved market competitiveness and client acquisition rates.
Conclusion
India's CDMO market is ready for a transformative growth, driven by regulatory support, technological advancements, and strategic investments. There are also predictions that this market will surpass global growth, driven by stringent regulations, tech innovations, and a skilled workforce.
Related tags
Get started
We've helped companies around the world future-proof
their businesses - and we can do the same for you.



















