
India Contract Manufacturing Organization (CMO) Market Outlook to 2028
Region:Asia
Author(s):Shambhavi
Product Code:KROD3195
November 2024
84
About the Report
India Contract Manufacturing Organization (CMO) Market Overview
- The India Contract Manufacturing Organization (CMO) market is valued at INR 1,200 billion, driven by the rising demand for generic pharmaceuticals and the robust growth of the biotechnology sector. This growth is also supported by favorable government policies encouraging foreign direct investment (FDI) in the healthcare and pharmaceutical industries. Additionally, India's cost-effective manufacturing capabilities, coupled with a highly skilled workforce, make it a key player in the global CMO market.
- In terms of regional dominance, key cities such as Hyderabad, Bengaluru, and Mumbai lead the Indian CMO market due to their well-established pharmaceutical manufacturing infrastructure and strong talent pools. These cities are home to several major pharmaceutical companies and have seen significant investment in R&D and biopharmaceutical production, contributing to their dominant position in the industry.

- Indian CMOs must comply with stringent drug manufacturing regulations to ensure product quality. In 2022, the CDSCO updated its Good Manufacturing Practices (GMP) guidelines, requiring pharmaceutical companies to adhere to higher standards of quality control. Over 1,000 companies underwent inspections, ensuring compliance with national and international standards. These regulations have helped Indian CMOs maintain a strong reputation for producing high-quality drugs.
India Contract Manufacturing Organization (CMO) Market Segmentation
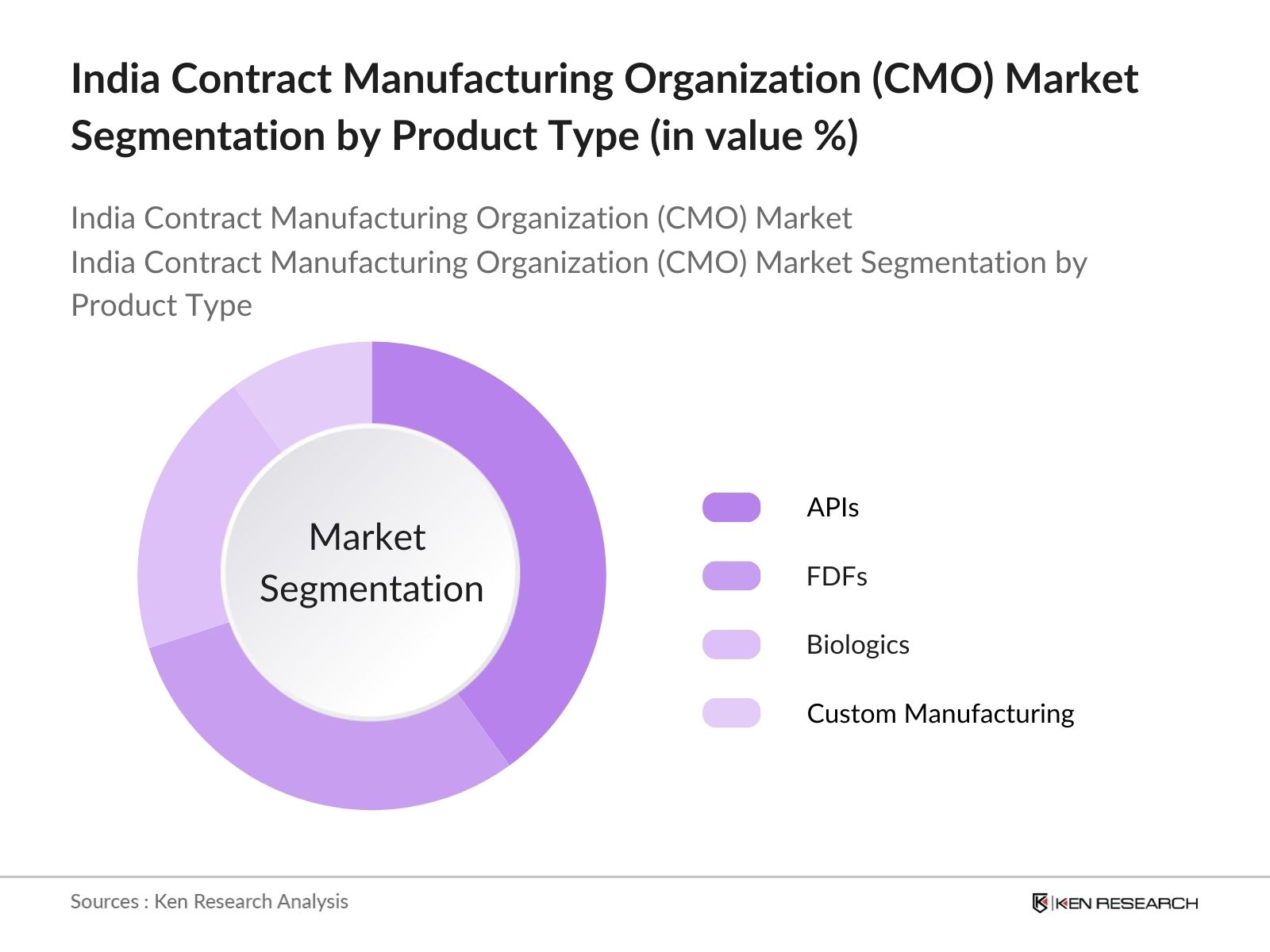
By Product Type:The India CMO market is segmented by product type into Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Biologics, and Custom Manufacturing. APIs currently hold the largest market share, primarily due to India's leadership in generic drug production and its cost-efficient manufacturing capabilities. This dominance is supported by India's large-scale production facilities and expertise in synthesizing complex molecules, which allows local manufacturers to supply APIs to both domestic and international markets.

By Service Type: The market is also segmented by service type, which includes Drug Development and Formulation, Analytical Services, Packaging and Labeling, and Clinical Manufacturing. Drug Development and Formulation dominate the market, as Indian CMOs have increasingly invested in R&D capabilities to offer comprehensive services to global pharmaceutical companies. This includes expertise in complex formulations, allowing Indian firms to serve a growing number of clients looking for end-to-end solutions from development to commercialization.
India Contract Manufacturing Organization (CMO) Market Competitive Landscape
The Indian CMO market is dominated by several major players who have established themselves as leaders through extensive service portfolios, global partnerships, and robust manufacturing capabilities. These companies are continuously expanding their service offerings to meet the increasing demand for contract manufacturing from both domestic and international pharmaceutical companies. The India CMO market is characterized by a few key players who have a significant influence on the market, including Cipla, Sun Pharma, and Biocon. These firms have leveraged their R&D capabilities, manufacturing infrastructure, and strategic partnerships to strengthen their market position.
|
Company Name |
Establishment Year |
Headquarters |
Manufacturing Facilities |
Product Portfolio |
R&D Spending |
Global Presence |
Certifications |
Strategic Partnerships |
|
Dr. Reddy's Laboratories |
1984 |
Hyderabad |
||||||
|
Cipla |
1935 |
Mumbai |
||||||
|
Biocon |
1978 |
Bengaluru |
||||||
|
Sun Pharma |
1983 |
Mumbai |
||||||
|
Aurobindo Pharma |
1986 |
Hyderabad |
India Contract Manufacturing Organization (CMO) Market Analysis
Growth Drivers
- Rising Demand for Generic Drugs: India is one of the largest suppliers of generic drugs globally, with around 20% of the world's generic medicines manufactured in the country. In 2023, generic drug exports from India reached USD 18.1 billion. The increasing healthcare demand worldwide, especially in developing nations, has led to a rise in outsourcing to Indian CMOs for the production of high-quality yet affordable generic drugs. This growth is supported by Indias extensive production base, skilled labor, and cost-effective manufacturing processes.
- FDI Policies Favoring Contract Manufacturing: India's government has implemented policies that attract Foreign Direct Investment (FDI) in the pharmaceutical sector, including 100% automatic approval in greenfield projects. In 2022, the pharmaceutical sector attracted FDI inflows worth USD 1.8 billion. This has encouraged international pharmaceutical companies to partner with Indian CMOs, providing a significant boost to contract manufacturing. FDI inflows help develop local manufacturing capacities and expand technological advancements in production, making India an attractive hub for outsourcing.
- Regulatory Approvals Streamlining: India has simplified its drug approval process to encourage contract manufacturing. The Central Drugs Standard Control Organization (CDSCO) has implemented faster regulatory pathways for drug approvals. In 2022, over 1,100 drugs received manufacturing approval, enabling CMOs to scale production quickly. Streamlined approvals and strong regulatory compliance have significantly contributed to the efficiency and growth of contract manufacturing in India, making it a favorable destination for global pharmaceutical companies.
Market Challenges
- Supply Chain Disruptions: Supply chain disruptions due to global geopolitical tensions and COVID-19 lockdowns have affected the pharmaceutical sector, causing delays in raw material supply. India relies heavily on imports of Active Pharmaceutical Ingredients (APIs), with approximately 68% sourced from China as of 2023. These disruptions have created challenges for Indian CMOs, leading to production delays and increased costs. The governments emphasis on boosting domestic API production through schemes like the PLI for Bulk Drugs aims to mitigate these risks.
- Strict Regulatory Requirements: While India is a preferred destination for contract manufacturing, navigating the regulatory frameworks of different countries remains a challenge. Indian CMOs face stringent international regulations, particularly from the US FDA and the European Medicines Agency. In 2022, around 42 Indian pharmaceutical companies faced FDA warnings, highlighting the complexity of adhering to global standards. This presents an ongoing challenge for Indian CMOs in maintaining compliance while scaling operations.
India Contract Manufacturing Organization (CMO) Market Future Outlook
Over the next five years, the India Contract Manufacturing Organization (CMO) market is expected to experience substantial growth, driven by increased outsourcing from global pharmaceutical companies, the rising demand for biologics and biosimilars, and favorable government policies that support pharmaceutical manufacturing. The expansion of manufacturing capacities, particularly for biopharmaceuticals, and the increasing focus on digital transformation and automation in production processes will also contribute to this growth.
Market Opportunities
- Expansion into Biologics Manufacturing: The growing global demand for biologics presents a significant opportunity for Indian CMOs to expand their services. As of 2023, biologics accounted for nearly 30% of global pharmaceutical revenues, with many large pharma companies looking to outsource their production. Indias burgeoning biopharmaceutical sector has received USD 200 million in government incentives through the PLI Scheme, encouraging local manufacturing of biologics and biosimilars. This expansion provides Indian CMOs with a pathway to enter higher-value manufacturing.
- Integration of Advanced Manufacturing Technologies: The integration of advanced technologies like artificial intelligence (AI), machine learning (ML), and automation is transforming the pharmaceutical manufacturing landscape. In 2024, India has seen increased adoption of digital manufacturing technologies, particularly among large CMOs. Government-backed initiatives such as the Digital India program have encouraged the use of smart manufacturing technologies. These advancements enhance production efficiency and reduce human error, providing Indian CMOs with a competitive edge.
Scope of the Report
|
Segment |
Sub-Segments |
|
By Product Type |
Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Biologics Manufacturing, Custom Manufacturing |
|
By Service Type |
Drug Development and Formulation, Analytical Services, Packaging and Labeling, Clinical Manufacturing |
|
By Application |
Oncology, Cardiovascular, Immunology, Neurology |
|
By End-User |
Big Pharma, Mid-sized Pharma, Biotech Companies |
|
By Region |
North India, South India, East India, West India |
Products
Key Target Audience
- Pharmaceutical Companies
- Biotechnology Firms
- Pharmaceutical Packaging Companies
- Healthcare Institutions
- Clinical Research Organizations (CROs)
- API and Excipient Suppliers
- Investors and Venture Capitalist Firms
- Government and Regulatory Bodies (CDSCO, Department of Pharmaceuticals)
Companies
Major Players mentioned in the Report
- Dr. Reddy's Laboratories
- Cipla
- Biocon
- Sun Pharma
- Aurobindo Pharma
- Lupin Pharmaceuticals
- Jubilant Life Sciences
- Zydus Cadila
- Piramal Pharma Solutions
- Divis Laboratories
- Hetero Labs
- Gland Pharma
- Laurus Labs
- Granules India
- Syngene International
Table of Contents
1. India Contract Manufacturing Organization Market Overview
1.1. Definition and Scope
1.2. Market Taxonomy
1.3. Market Growth Rate
1.4. Market Segmentation Overview
2. India Contract Manufacturing Organization Market Size (In INR Bn)
2.1. Historical Market Size
2.2. Year-On-Year Growth Analysis
2.3. Key Market Developments and Milestones
3. India Contract Manufacturing Organization Market Analysis
3.1. Growth Drivers
3.1.1. Pharmaceutical Outsourcing (Market Specific Parameter)
3.1.2. Rising Demand for Generic Drugs (Market Specific Parameter)
3.1.3. FDI Policies Favoring Contract Manufacturing (Market Specific Parameter)
3.1.4. Regulatory Approvals Streamlining (Market Specific Parameter)
3.2. Market Challenges
3.2.1. Supply Chain Disruptions (Market Specific Parameter)
3.2.2. Strict Regulatory Requirements (Market Specific Parameter)
3.2.3. Price Pressures from Global Competition (Market Specific Parameter)
3.3. Opportunities
3.3.1. Expansion into Biologics Manufacturing (Market Specific Parameter)
3.3.2. Integration of Advanced Manufacturing Technologies (Market Specific Parameter)
3.3.3. Growing Partnerships with Global Pharma Firms (Market Specific Parameter)
3.4. Trends
3.4.1. Rise of Digital Manufacturing and Automation (Market Specific Parameter)
3.4.2. Increase in Biopharmaceutical Outsourcing (Market Specific Parameter)
3.4.3. Focus on Sustainable Manufacturing Practices (Market Specific Parameter)
3.5. Government Regulations
3.5.1. Drug Manufacturing Compliance Regulations (Market Specific Parameter)
3.5.2. Import-Export Policies for Pharma APIs (Market Specific Parameter)
3.5.3. PLI (Production Linked Incentive) Scheme for Pharmaceuticals (Market Specific Parameter)
3.6. SWOT Analysis
3.7. Stakeholder Ecosystem
3.8. Porters Five Forces
3.9. Competition Ecosystem
4. India Contract Manufacturing Organization Market Segmentation
4.1. By Product Type (In Value %)
4.1.1. Active Pharmaceutical Ingredients (APIs)
4.1.2. Finished Dosage Forms (FDFs)
4.1.3. Biologics Manufacturing
4.1.4. Custom Manufacturing
4.2. By Service Type (In Value %)
4.2.1. Drug Development and Formulation
4.2.2. Analytical Services
4.2.3. Packaging and Labeling
4.2.4. Clinical Manufacturing
4.3. By Application (In Value %)
4.3.1. Oncology
4.3.2. Cardiovascular
4.3.3. Immunology
4.3.4. Neurology
4.4. By End-User (In Value %)
4.4.1. Big Pharma
4.4.2. Mid-sized Pharma
4.4.3. Biotech Companies
4.5. By Region (In Value %)
4.5.1. North India
4.5.2. South India
4.5.3. East India
4.5.4. West India
5. India Contract Manufacturing Organization Market Competitive Analysis
5.1. Detailed Profiles of Major Companies
5.1.1. Dr. Reddys Laboratories
5.1.2. Cipla
5.1.3. Biocon
5.1.4. Piramal Pharma Solutions
5.1.5. Sun Pharma
5.1.6. Lupin Pharmaceuticals
5.1.7. Aurobindo Pharma
5.1.8. Zydus Cadila
5.1.9. Jubilant Life Sciences
5.1.10. Syngene International
5.1.11. Gland Pharma
5.1.12. Divis Laboratories
5.1.13. Hetero Labs
5.1.14. Laurus Labs
5.1.15. Granules India
5.2. Cross Comparison Parameters (No. of Employees, Headquarters, Product Portfolio, Manufacturing Capacity, Revenue, Market Presence, R&D Spending, Regulatory Approvals)
5.3. Market Share Analysis
5.4. Strategic Initiatives
5.5. Mergers And Acquisitions
5.6. Investment Analysis
5.7. Venture Capital Funding
5.8. Government Grants
5.9. Private Equity Investments
6. India Contract Manufacturing Organization Market Regulatory Framework
6.1. Drug Manufacturing Compliance Standards
6.2. Compliance Requirements for Biologics
6.3. API Import Regulations
6.4. Licensing and Certification Processes
7. India Contract Manufacturing Organization Future Market Size (In INR Bn)
7.1. Future Market Size Projections
7.2. Key Factors Driving Future Market Growth
8. India Contract Manufacturing Organization Future Market Segmentation
8.1. By Product Type (In Value %)
8.2. By Service Type (In Value %)
8.3. By Application (In Value %)
8.4. By End-User (In Value %)
8.5. By Region (In Value %)
9. India Contract Manufacturing Organization Market Analysts Recommendations
9.1. TAM/SAM/SOM Analysis
9.2. Market Opportunity Analysis
9.3. Customer Segmentation
9.4. Strategic Partnerships
Disclaimer
Contact Us
Research Methodology
Step 1: Identification of Key Variables
The first step involves mapping the entire ecosystem of the India CMO market. This is achieved through extensive desk research, using both secondary sources such as industry reports and proprietary databases. The objective is to identify the major factors influencing market dynamics, such as demand drivers, regulatory frameworks, and emerging trends.
Step 2: Market Analysis and Construction
In this phase, we assess historical data related to the markets performance. This includes analyzing contract manufacturing growth, the relationship between service providers and pharmaceutical companies, and market revenue generated. Additionally, a review of service quality and output statistics is conducted to ensure the accuracy of market projections.
Step 3: Hypothesis Validation and Expert Consultation
We develop market hypotheses that are then validated through interviews with industry experts, including professionals from leading pharmaceutical companies. These consultations provide insights into operational trends and financial data, ensuring that our market estimates are accurate and reliable.
Step 4: Research Synthesis and Final Output
In the final step, we engage with major manufacturers and service providers in the CMO sector to gain in-depth insights into product segments, customer preferences, and emerging market trends. This helps validate the research and ensures the delivery of a comprehensive and accurate market report.
Frequently Asked Questions
01. How big is the India Contract Manufacturing Organization (CMO) market?
The India Contract Manufacturing Organization market is valued at INR 1,200 billion, driven by rising demand for generic pharmaceuticals, biologics, and biosimilars.
02. What are the challenges in the India CMO market?
Key challenges in India Contract Manufacturing Organization Market include supply chain disruptions, stringent regulatory requirements, and pricing pressures from global competition, which create operational complexities for manufacturers.
03. Who are the major players in the India CMO market?
Key players in India Contract Manufacturing Organization Market include Dr. Reddy's Laboratories, Cipla, Biocon, Sun Pharma, and Aurobindo Pharma. These companies dominate due to their strong manufacturing capabilities, R&D investments, and global partnerships.
04. What are the growth drivers of the India CMO market?
The India Contract Manufacturing Organization market is propelled by increasing outsourcing of pharmaceutical production, rising demand for biopharmaceuticals, and favorable government policies that support pharmaceutical manufacturing.
Why Buy From Us?

What makes us stand out is that our consultants follows Robust, Refine and Result (RRR) methodology. i.e. Robust for clear definitions, approaches and sanity checking, Refine for differentiating respondents facts and opinions and Result for presenting data with story

We have set a benchmark in the industry by offering our clients with syndicated and customized market research reports featuring coverage of entire market as well as meticulous research and analyst insights.

While we don't replace traditional research, we flip the method upside down. Our dual approach of Top Bottom & Bottom Top ensures quality deliverable by not just verifying company fundamentals but also looking at the sector and macroeconomic factors.

With one step in the future, our research team constantly tries to show you the bigger picture. We help with some of the tough questions you may encounter along the way: How is the industry positioned? Best marketing channel? KPI's of competitors? By aligning every element, we help maximize success.

Our report gives you instant access to the answers and sources that other companies might choose to hide. We elaborate each steps of research methodology we have used and showcase you the sample size to earn your trust.

If you need any support, we are here! We pride ourselves on universe strength, data quality, and quick, friendly, and professional service.















